TraC is a miniature handheld instrument for measuring trace chemical contaminants on surfaces, especially focused on cleaning validation and process control for food or pharmaceutical product manufacturing.
The TraC sensor has a ppb limit of detection for many chemical & biological materials on surfaces as well as small concentrations of chemical and microbial material in liquids. It also has a large depth of focus.
In its present configuration, TraC has an internal microprocessor to control all sensor functions and to process all spectral data to determine concentrations of targeted materials on surfaces at ppb limits of detection in << 1 s. However, in the case of pharma cleaning validation, the raw data is presently communicated with the pharma user for analysis for legal and FDA reasons.
TraC weighs less than 2lbs and can operate for over 24 hours on an internal battery without recharge. TraC can also be fitted with liquid samplers, flow cells, or windows to look directly into mixing or bioreactor growth chambers or to inspect manufactured products or production surfaces for product quality control or equipment cleaning validation. TraC is compatible with Photon Systems’ ChemCal XY printing, mapping and calibration system described here.


Key Features
Real Time Optical Scanning
Rapidly finds hot spots for further testing
High Sensitivity
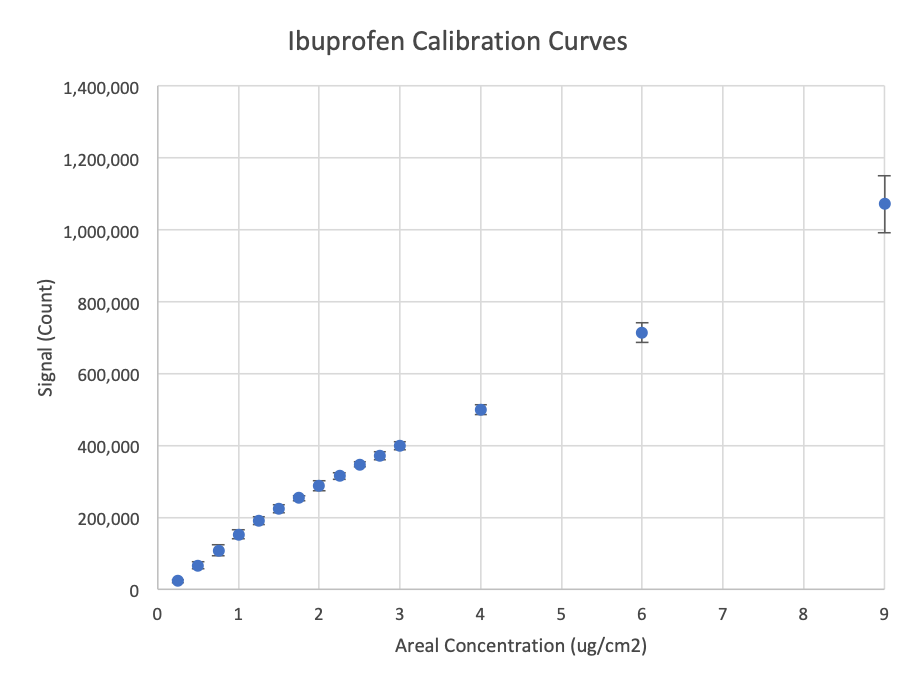
< 1 ug/cm2 Limit of Detection
High Selectivity
>95% differentiability of APIs, excipients, etc.
Non-Contact
Working distance of 0.5 to 2.0 cm
Downloads
ChemCal
A trace chemical detector like the TraC is acceptable for P.A.T. and many other applications only if you can accurately calibrate its performance and generate reproducible chemical concentration curves. That is why we created the ChemCal.
Pharmaceutical Cleaning Validation
Detecting and quantifying trace residue concentrations of active pharmaceutical ingredients (API), excipients, and washes during cleaning validation procedures on pharmaceutical manufacturing equipment is one of the single largest costs associated with the manufacture of pharmaceutical drugs.
Food Equipment Cleaning Validation
Traditional methods of food equipment cleaning validation include microbial assays and visual inspection. However, these techniques have limitations. Using deep UV fluorescence techniques, Photon Systems handheld detectors are capable of detecting trace organic residues as low as pg/cm2 and biological contamination less than hundreds of cells or spores.
Get In Touch
Keep informed about the latest deep UV developments at Photon Systems by joining our mailing list.
ADDRESS
1512 Industrial Park St. Covina, CA 91722-3417
PHONE
626 967-6431




